A few experiments
Patent blue V
To begin we did a very simple experiment that allows to measure the absorbance of solutions containing different known concentrations of patent blue V (E131), a blue pigment that is used in various blue candies.
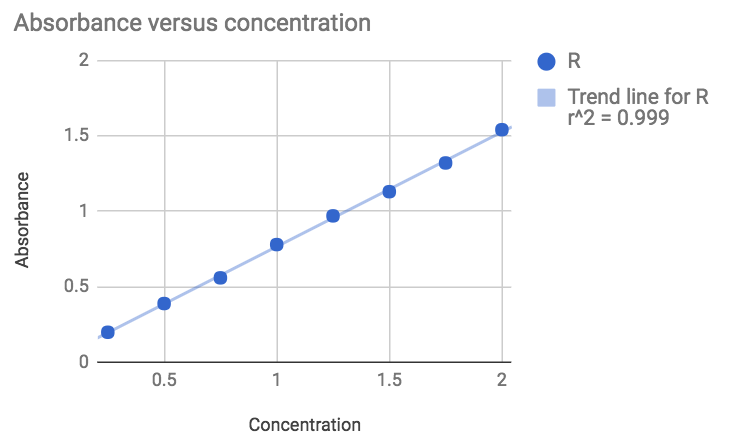
The result is rather good knowing that the solutions were simply prepared by adding various volumes (0 to 2 mL) of a concentrated solution with a 1mL seringe to 100mL of water. Not the mst accurate method.
You may check the following protocols
Fluorescence quenching
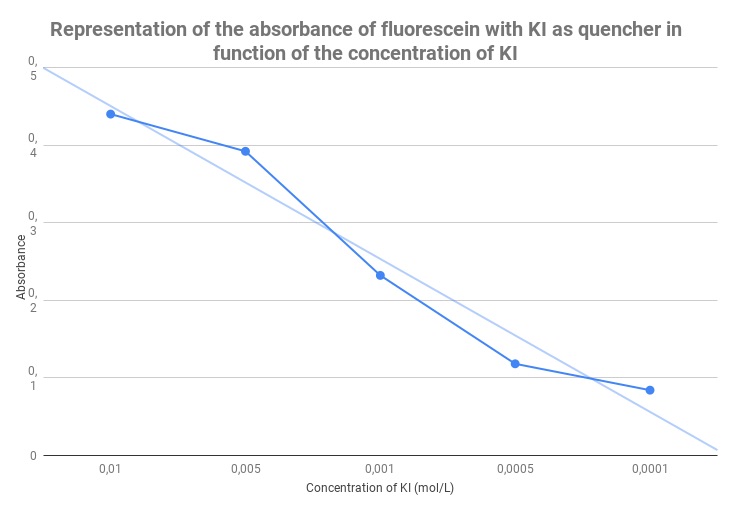
In order to determine if the spectrophotometer is able to measure the difference of UV absorbance in a fluorescence quenching situation, we used fluorescein and potassium iodide (KI) as a quencher.
To do it, we started with a solution of KI 0.01M that was diluted to obtain solutions at 0.005M, 0.001M, 0.0005M, 0.0001M. Then from each solution we picked 3 drops and put them in a tank full of florescein (concentration?!).
Here are the results:
KI 0.01M:
| MEAN | SD | |
|---|---|---|
| UV absorbance | 0,44 | 0,021 |
KI 0.005M:
| MEAN | SD | |
|---|---|---|
| UV absorbance | 0,392 | 0,019 |
KI 0.001M:
| MEAN | SD | |
|---|---|---|
| UV absorbance | 0,232 | 0,022 |
KI 0.0005M:
| MEAN | SD | |
|---|---|---|
| UV absorbance | 0,118 | 0,018 |
KI 0.0001M:
| MEAN | SD | |
|---|---|---|
| UV absorbance | 0,084 | 0,011 |
Original data
If you want to see all our measurements, please click on the following link:
Detection of mercury water pollution using carbon quantum dots
In order to detect mercury, carbon quantum dots (CQDs) can be used. They are small particles of carbon (which can be homemade) that become fluorescent in presence of UV light. The interesting part of this application is that the CQDs react differently with mercury, and that this difference can be measured using the simple-spectrophotometer.

How to make CQDs ?
Materials
– 14g of sucrose or fructose
– 40ml of lemon juice or 40 ml of water and 20 drops of HCl 18%
– 0.5 g of sodium bicarbonate
– a hot plate
– a magnetic stirrer
– an aluminum foil
– a beaker
– a spatula
Procedure
- Mix the sucrose/ fructose with your chosen acidic solution (we used an erlenmeyer), using the magnetic agitator.
- Heat your solution up to 250°C thanks to the hot plate, for 40 minutes until it becomes black. Be careful to cover (with the aluminum foil) and agitate your solution.
- Pour the content in a beaker and add in two times the sodium bicarbonate (your mix will inflate).
- If the mixture is too thick, don't hesitate to add up to 10ml of water.
- Let cool down the solution of CQDs.
To use the CQDs, you will have to dilute them (for more precise quantities, please wait, work in progress...!)
Test of fluorescence
Using the simple-spectrophotometer, we tested for the reaction of CQDs when exposed to UV light, while the particles were diluted in water. We used water as blank, and test for different concentration of CQDs (1, 3 or 5 drops per tank). We obtained those results:
One drop of CQDs
| MEAN | Blank | Energy | Absorbance | SD | Blank | Energy | Absorbance | |
|---|---|---|---|---|---|---|---|---|
| R | 395119,3 | 368277,9 | 0,031 | 5418,35 | 7564,05 | 0,011 | ||
| G | 419962,1 | 293356,4 | 0,157 | 1908,28 | 16532,62 | 0,027 | ||
| B | 473191,5 | 217507,5 | 0,34 | 4166,15 | 25876,94 | 0,055 | ||
| UV | 6548,1 | 4920,6 | 0,124 | 179,36 | 224,38 | 0,020 |
Three drops of CQDs
| MEAN | Blank | Energy | Absorbance | SD | Blank | Energy | Absorbance | |
|---|---|---|---|---|---|---|---|---|
| R | 389990,3 | 296282,5 | 0,115 | 6210,02 | 12258,36 | 0,027 | ||
| G | 415605,1 | 147389,4 | 0,457 | 11265,94 | 17618,44 | 0,052 | ||
| B | 476379 | 49219,3 | 0,997 | 5033,65 | 12225,96 | 0,11 | ||
| UV | 6683,5 | 4221 | 0,2 | 170,83 | 147,41 | 0,019 |
Five drops of CQDs
| MEAN | Blank | Energy | Absorbance | SD | Blank | Energy | Absorbance | |
|---|---|---|---|---|---|---|---|---|
| R | 391031,3 | 241988,8 | 0,209 | 5801,56 | 10126,13 | 0,024 | ||
| G | 418085,4 | 78945,4 | 0,73 | 7647,39 | 10989,91 | 0,062 | ||
| B | 468810,2 | 13237,1 | 1,57 | 18055,30 | 3902,91 | 0,12 | ||
| UV | 6912,3 | 4021,5 | 0,234 | 202,21 | 123,86 | 0,016 |
Plotting the absorbance of the samples, we obtain the following graph:
The R squared is equal to 0.958.
You can find all the values here:
Determination of the pH using red cabbage
The color of the red cabbage highly depends on the pH of a solution. We are trying to determine if it is possible to create a model with the simple-spectrophotometer to allow the users to estimate easily the pH of their solution.
How to create your red cabbage solution ?
Materials
– 500g of red cabbage
Procedure
- Cut your red cabbage in small pieces.
- Boil it in 1L of tap water for ~ 25 min.
- Collect the juice and store it in the fridge.
We are trying to create a precise procedure to use it, for the moment you can see the results of our attempts: